lec 5 (PDF)
File information
Title: Slide 1
Author: Administrator
This PDF 1.5 document has been generated by Microsoft® PowerPoint® 2010, and has been sent on pdf-archive.com on 06/12/2017 at 23:55, from IP address 197.196.x.x.
The current document download page has been viewed 352 times.
File size: 678.24 KB (30 pages).
Privacy: public file



File preview
Alexandria University
Faculty of Engineering
Department of Chemical Engineering
CH211: Organic Chemistry 3
5th lecture
Fall 2017
CH211:Organic Chemistry 3
Dr. Iman El Gheriany
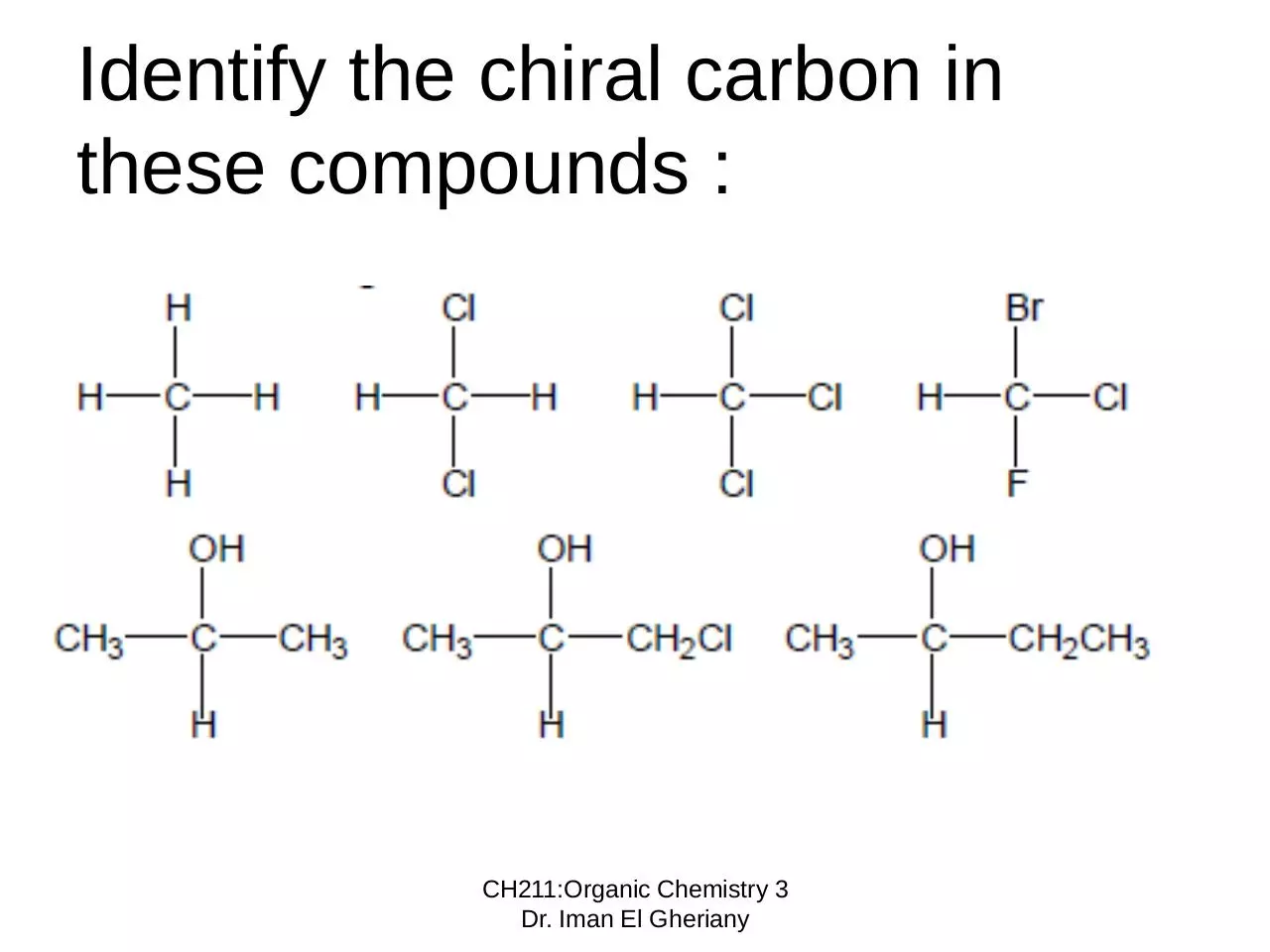
Identify the chiral carbon in
these compounds :
CH211:Organic Chemistry 3
Dr. Iman El Gheriany
TEST YOURSELF QUESTIONS
1-Write down the Fischer Projection of the product you get by the Kiliani-Fischer
synthesis of D-ribose?
CH211:Organic Chemistry 3
Dr. Iman El Gheriany
CH211:Organic Chemistry 3
Dr. Iman El Gheriany
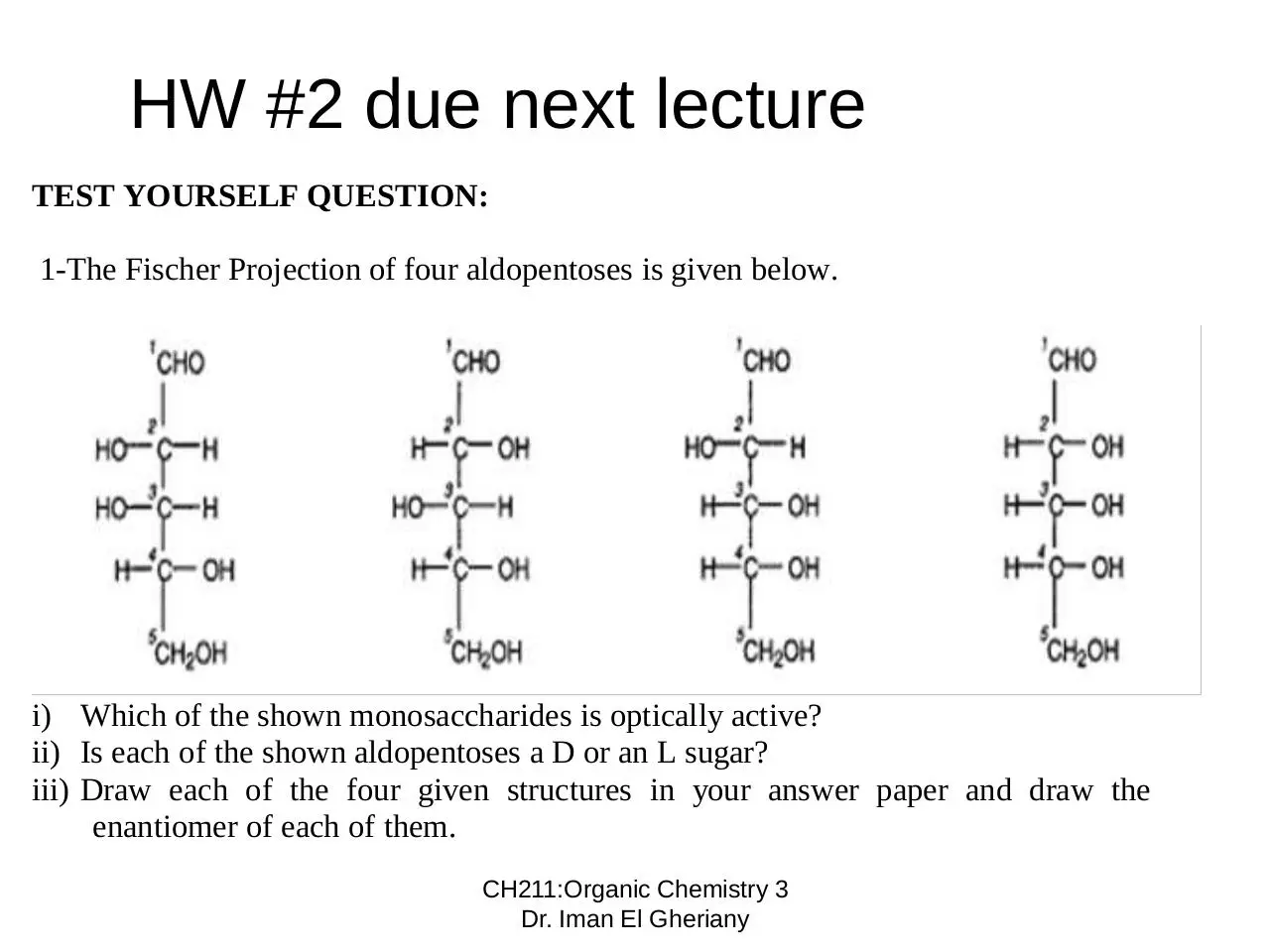
HW #2 due next lecture
TEST YOURSELF QUESTION:
1-The Fischer Projection of four aldopentoses is given below.
i) Which of the shown monosaccharides is optically active?
ii) Is each of the shown aldopentoses a D or an L sugar?
iii) Draw each of the four given structures in your answer paper and draw the
enantiomer of each of them.
CH211:Organic Chemistry 3
Dr. Iman El Gheriany
Which of the eight D-aldohexoses yield
optically inactive (meso) aldaric acids on
oxidation?
CH211:Organic Chemistry 3
Dr. Iman El Gheriany
Which D-aldohexoses give the same ozasone ?
CH211:Organic Chemistry 3
Dr. Iman El Gheriany
What aldopentose would give a mixture of L-gulose
and L-idose on Kiliani-Fischer chain extension
SUPPLEMENT
Download lec 5
lec 5.pdf (PDF, 678.24 KB)
Download PDF
Share this file on social networks
Link to this page
Permanent link
Use the permanent link to the download page to share your document on Facebook, Twitter, LinkedIn, or directly with a contact by e-Mail, Messenger, Whatsapp, Line..
Short link
Use the short link to share your document on Twitter or by text message (SMS)
HTML Code
Copy the following HTML code to share your document on a Website or Blog
QR Code to this page

This file has been shared publicly by a user of PDF Archive.
Document ID: 0000705354.